What is Gas chromatography?
Gas chromatography is a physical analysis technique using gas as mobile phase and column chromatography.

Column of Gas Chromatography
The chromatographic column is the core component of gas chromatography, and its quality plays an important role in the indicators of the entire gas chromatography. There are two types of chromatographic columns: packed element and capillary column. The packed column is filled with a solid adsorbent or a carrier with a fixed liquid, and different packings can be selected to separate various compounds with high boiling points such as nitrogen, oxygen, hydrogen, and Ar. The capillary column has high separation efficiency, and the stationary phase liquid is coated on the inner wall of the capillary with a small inner diameter. Stationary phases can be divided into solid stationary phases and liquid stationary phases. The solid stationary phase is an adsorbent with different adsorption capacities for different components. For example, sucrose, starch, inulin, talc, potassium carbonate, etc. are weak adsorbents, and their adsorption is enhanced in turn.
Outstanding Advantages of Gas Chromatography
1. High separation efficiency. Complex mixed substances with very close physical and chemical properties can be well separated, and qualitative and quantitative detection can be carried out. Sometimes tens or even hundreds of components can be resolved simultaneously in one analysis.
2. High sensitivity. The impurity content of ppm level or even ppb level can be detected, and only a gas sample of less than 1mL or a liquid sample of less than 1 is required.
3. The analysis speed is fast. Thanks to the computer, accurate analysis results can be obtained even in seconds.
4. Wide range of applications. Gas chromatography can analyze gaseous, volatile liquid, and solid samples. As far as organic analysis is concerned, it is the most widely used and can analyze about 20% of organics. In addition, some inorganic substances can also be analyzed by transformation.
General procedure for a gas chromatography
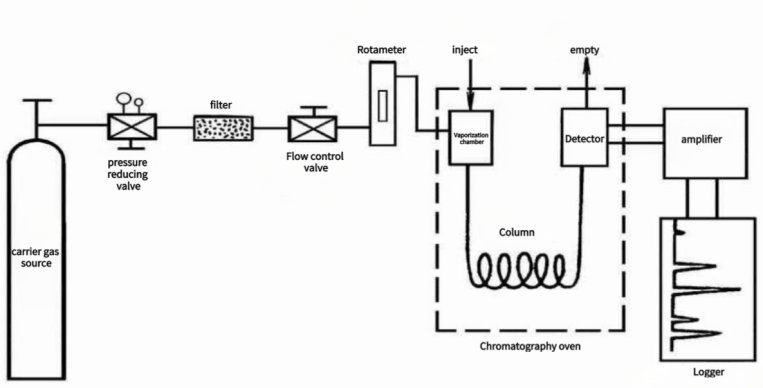
General procedure for a gas chromatography
Separation principle of a chromatographic column
The sequence of peak times can be used as the basis for qualitative analysis, and the peak area or peak height can be used as the basis for quantitative analysis.
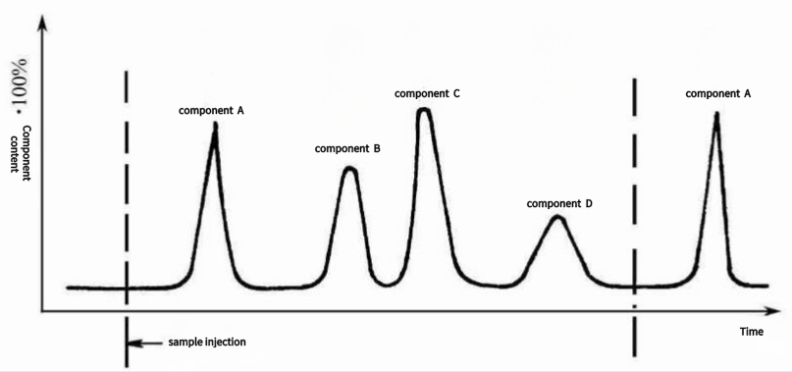
Working principle of a chromatographic
Three Components of an Industrial Gas Chromatography
Compared with general laboratory gas chromatography, the sampling system is added in industrial gas chromatography. The column-cutting technology is used, and the program control and information processing are completely automated.
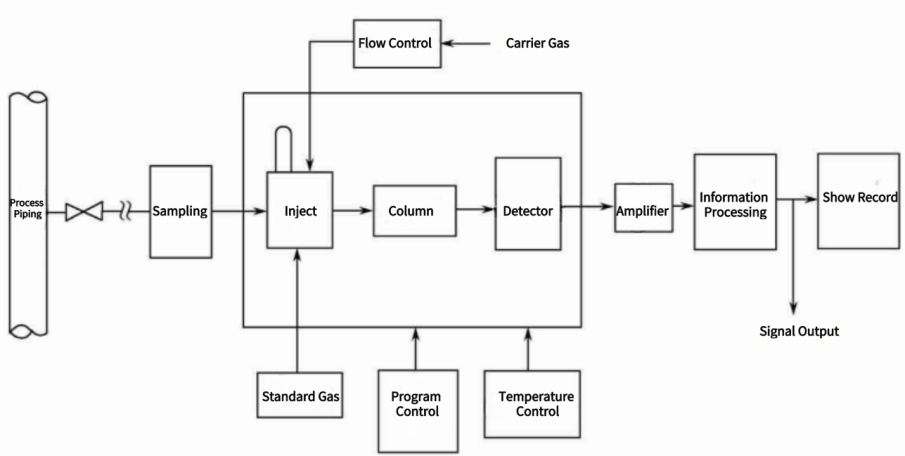
1. Sampling system
The sampling system completes the tasks of sampling and sample pretreatment and is the interface equipment of the production device and the industrial gas chromatography. The initial pretreatment of the sample should be carried out outside the gas chromatography, such as decompression, water removal, dust removal, etc.
The internal sampling and sample pretreatment system of the gas chromatography should have functions such as pressure regulation, flow path switching, flow monitoring, atmospheric balance, and standard gas (or standard liquid) calibration. Other practical issues should be considered in the design of sampling and sample preparation systems, such as corrosion resistance of system piping and components, prevention of leaks, explosion protection, reduction of transfer lag time, and control of contamination from evacuation.
2. Carrier gas flow system
The carrier gas flow system of gas chromatography includes a carrier gas source, purifier, pressure and flow stabilization, and adjustment devices. Usually, a high-pressure gas in a cylinder is used as the carrier gas source. There should be a pressure-reducing valve at the outlet of the cylinder to reduce the pressure to 0.1~0.5MPa. To avoid contamination of the chromatographic column, the carrier gas is required to be of high purity and good stability. Therefore, silica gel, molecular sieve, and activated carbon are often used to adsorb moisture and hydrocarbon compounds in the carrier. Gases that can be used as carrier gas include hydrogen, nitrogen, argon, and the like.
Gas chromatography requires that the carrier gas flow be kept constant and its variation should be less than 1%. Therefore, a flow meter and a regulating valve should be configured in the gas path, and a pressure-stabilizing valve should be installed to continuously adjust and stabilize the air pressure of the flow path, to achieve the purpose of stabilizing the flow.
3. Sampling device
Sampling is the quantitative addition of a gas, liquid, or converted solid sample to the head of a chromatographic column for chromatographic separation. The constancy of the number of injections, the length of injection time, and the speed of sample vaporization will affect the repeatability and accuracy of quantitative results.
(1). Gasification chamber
The outer wall of the gasification chamber is made of metal blocks, and the working temperature can be controlled between 50-500 °C. When the working temperature is higher than 250 ℃, to prevent the positive catalytic effect, it is advisable to adopt the structure of the inner glass tube.
The function of the gasification chamber:
It is ensured that the liquid sample vaporizes instantaneously in it. The carrier gas should be preheated before entering the vaporizer, but the silicone rubber pad should be cooled to avoid unwanted chemical reactions.
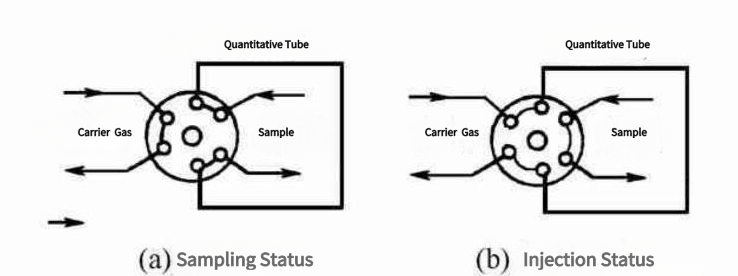
(2). Injection valve
Sampling valve requirements: good air tightness, small dead volume, reliable and durable, fast switching time. On some occasions, it is also required to be able to resist corrosion and work under certain temperature conditions.
In conclusion, to know more about gas chromatography, please contact the gas chromatography manufacturer.






